Screening for p53 would predict sensitivity to proteasome inhibitors
Mar 10, 2007 - 11:40:03 AM
, Reviewed by: Dr. Rashmi Yadav
|
|
|
If confirmed experimentally, our hypothesis will serve to pre-select patients with the best chances of success those with p53 and spare the rest those without p53 the severe side effects of bortezomib therapy."
|
|
Key Points of this article
|
|
Researchers at the University of Pennsylvania Schools of Medicine and Veterinary Medicine propose to test for p53, a well-known tumor-suppressor protein, in cancer patients to determine if they are suitable candidates for proteasome inhibitors, a promising class of anti-cancer drugs.
|
|
The researchers speculate that responsive myelomas are the ones retaining the p53 protein, which gets stabilized during treatment and triggers self-destruction of cancerous cells.
|
|
|
|
By University of Pennsylvania School of Medicine,
[RxPG]
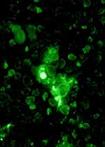 |
| Green staining depicts tumor cells dying after treatment with proteasome inhibitors. Here, the tumor lacks p53, and only a fraction of the cells are killed.
|
Researchers at the University of Pennsylvania Schools of Medicine and Veterinary Medicine have determined a way to pre-screen cancer patients to see if they are suitable candidates for proteasome inhibitors, a promising class of anti-cancer drugs. They propose to test for p53, a well-known tumor-suppressor protein that is broken down by cellular machinery called proteasomes. This study appears online in the journal Blood, in advance of print publication in June 2007.
In cancer patients whose tumors do not produce p53, proteasome inhibitors might be ineffective. This patient group could be spared unnecessary treatment with possible harmful side effects. On the other hand, proteasome inhibitors are highly effective against lymphomas that do have the ability to produce p53.
Proteasomes resemble paper shredders they break down proteins such as p53 into smaller pieces, says senior author Andrei Thomas-Tikhonenko, PhD, Associate Professor of Pathology. A proteosome inhibitor effectively jams the shredder so that p53 is not immediately broken down.
In this study, the research team used a mutant strain of mice in which p53 activity can be switched on and off. In principle, tumors in these mice could be obliterated by turning p53 back on, says Thomas-Tikhonenko. The problem was that a protein called MDM2 sent p53 into the teeth of the proteasome shredder. The proteasome inhibitor bortezomib (Velcade®) causes this jamming process and restores p53 function. However, if p53 was inactivated in the mice, bortezomib treatment failed to kill tumors. Similar effects were seen with cell lines derived from human Burkitts lymphomas. When implanted into mice, these lymphoma cells were highly sensitive to the proteasome inhibitor, but as soon as p53 was removed, the inhibitor had no effect.
These findings have important implications for clinical practice, Thomas-Tikhonenko adds. Bortezomib is approved by the Food and Drug Administration for the treatment of multiple myeloma, another cancer of lymphoid cells. Yet, only a fraction of multiple myeloma patients respond to the drug.
The researchers speculate that responsive myelomas are the ones retaining the p53 protein, which gets stabilized during treatment and triggers self-destruction of cancerous cells. If confirmed experimentally, our hypothesis will serve to pre-select patients with the best chances of success those with p53 and spare the rest those without p53 the severe side effects of bortezomib therapy, explains Thomas-Tikhonenko.
There are two ways, suggest the researchers, to test for p53 production in cancer patients. First, if MDM2 is expressed at abnormally high levels, it is a good indicator that p53 is constantly being made. Second, genetic tests can also be conducted to see if the malignant cells still have the gene for p53 or if the portion of the chromosome on which p53 is found has been deleted.
In addition to Thomas-Tikhonenko, Duonan Yu and Martin Carroll, both from Penn, were co-authors. The National Institutes of Health and the Commonwealth of Pennsylvania Health Research Formula Fund provided funding for this research.
Advertise in this space for $10 per month.
Contact us today.
|
 |
|
Subscribe to Blood Newsletter
|
|
|
|
About Dr. Rashmi Yadav
|
This news story has been reviewed by Dr. Rashmi Yadav before its publication on RxPG News website. Dr. Rashmi Yadav, MBBS, is a senior editor for RxPG News. In her position she is responsible for managing special correspondents and the surgery section of the website. Her areas of special interest include cardiothoracic surgery and interventional radiology.
RxPG News is committed to promotion and implementation of Evidence Based Medical Journalism in all channels of mass media including internet.
|
|
Additional information about the news article
|
PENN Medicine is a $2.9 billion enterprise dedicated to the related missions of medical education, biomedical research, and high-quality patient care. PENN Medicine consists of the University of Pennsylvania School of Medicine (founded in 1765 as the nation's first medical school) and the University of Pennsylvania Health System.
Penn's School of Medicine is ranked #2 in the nation for receipt of NIH research funds; and ranked #3 in the nation in U.S. News & World Report's most recent ranking of top research-oriented medical schools. Supporting 1,400 fulltime faculty and 700 students, the School of Medicine is recognized worldwide for its superior education and training of the next generation of physician-scientists and leaders of academic medicine.
The University of Pennsylvania Health System includes three hospitals, all of which have received numerous national patient-care honors [Hospital of the University of Pennsylvania; Pennsylvania Hospital, the nation's first hospital; and Penn Presbyterian Medical Center]; a faculty practice plan; a primary-care provider network; two multispecialty satellite facilities; and home care and hospice.
|
|
Feedback
|
For any corrections of factual information, to contact the editors or to send
any medical news or health news press releases, use
feedback form
|
Top of Page
|



